Home /
Expert Answers /
Chemistry /
question-4-1-pts-two-solutions-the-system-each-with-a-volume-of-25-0-ml-at-25-0-deg-c-are-mixed-pa454
(Solved): Question 4 1 pts Two solutions (the system), each with a volume of 25.0 mL at 25.0\deg C, are mixed ...
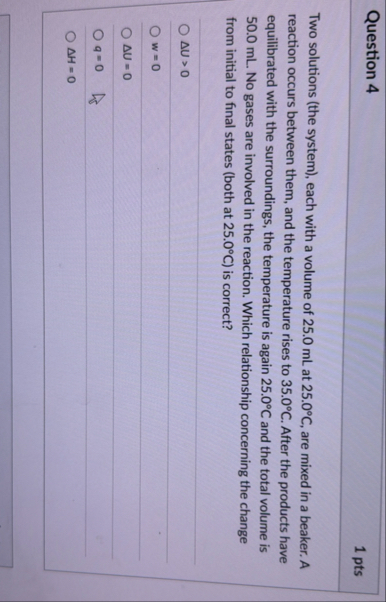
Question 4 1 pts Two solutions (the system), each with a volume of 25.0 mL at
25.0\deg C, are mixed in a beaker. A reaction occurs between them, and the temperature rises to
35.0\deg C. After the products have equilibrated with the surroundings, the temperature is again
25.0\deg Cand the total volume is 50.0 mL . No gases are involved in the reaction. Which relationship concerning the change from initial to final states (both at
25.0\deg C) is correct?
?
\Delta U>0
w=0
\Delta U=0
?
q=0
?
\Delta H=0